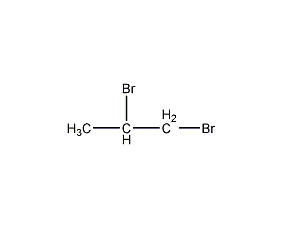
Structural formula
| Business number | 01NF |
|---|---|
| Molecular formula | C3H6Br2 |
| Molecular weight | 201.89 |
| label |
Dibromopropane, propylene dibromide, 2-bromopropyl bromide, Propylene dibromide, Propylene bromide, Aliphatic halogenated derivatives |
Numbering system
CAS number:78-75-1
MDL number:MFCD00000148
EINECS number:201-139-1
RTECS number:TX8574000
BRN number:1718884
PubChem number:24848483
Physical property data
1. Properties: colorless to yellow liquid [1]
2. Melting point (℃): -55.2[2 ]
3. Boiling point (℃): 140~142[3]
4. Relative density (water=1): 1.94 [4]
5. Saturated vapor pressure (kPa): 1.33 (35.7℃) [5]
6. Critical pressure (MPa): 4.08[6]
7. Octanol/water partition coefficient: 2.43[7]
8. Flash point (℃): <-75[8]
9. Solubility: insoluble in water, miscible in ethanol, ether, acetone, and chloroform. [9]
10. Viscosity (mPa·s, 20ºC): 1.62
11. Relative evaporation rate (ether=1): 28
12. Solubility (%, water, 20ºC): 0.25
13. Relative density (20℃, 4℃): 1.9323
14. Relative density (25 ℃, 4℃): 1.9420
15. Solubility parameter (J·cm-3)0.5: 20.852
16.van der Waals area (cm2·mol-1): 8.200×109
17.van der Waals volume (cm3·mol-1): 60.100
18. The gas phase standard claims heat (enthalpy) (kJ·mol -1): -71.5
19. Liquid phase standard claims heat (enthalpy) (kJ·mol-1): -113.2
20. Liquid phase standard hot melt (J·mol-1·K-1): 160.6
Toxicological data
1. Acute toxicity[10]
LD50: 741mg/kg (rat oral)
LC50: 12000mg/m3 (rat inhalation, 4h)
2. Irritation No information available
Ecological data
1. Ecotoxicity No data available
2. Biodegradability No data available
3. Non-biodegradability[11] In the air, when the concentration of hydroxyl radicals is 5.00×105/cm When 3, the degradation half-life is 37 days (theoretical).
At 25℃, when the pH value is 7, the hydrolysis half-life is 320d (theoretical).
Molecular structure data
1. Molar refractive index: 31.36
2. Molar volume (cm3/mol): 104.7
3. Isotonic specific volume (90.2K ): 253.1
4. Surface tension (dyne/cm): 34.0
5. Polarizability: 12.43
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 2.2
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 0
4. Number of rotatable chemical bonds: 1
5. Number of tautomers: none
6. Topological molecule polar surface area 0
7. Number of heavy atoms: 5
8. Surface charge: 0
9. Complexity: 20.9
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of atomic stereocenters: 1
13. Determine the number of chemical bond stereocenters: 0
14. Number of uncertain chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
1. This product is a non-flammable and non-explosive liquid. The solubility in water is 0.2% at 25°C and 0.3% at 80°C. It can decompose and release poisonous gas when exposed to high temperatures. Does not burn or explode. When heated, part of it becomes 1,3-dibromopropane. Substitution and addition reactions can occur. The decomposition temperature is about 425℃. Pay attention to ventilation and protection during production and use.
2. Stability[12] Stable
3. Incompatible substances[13] Strong alkali, strong oxidizing agent, magnesium
4. Conditions to avoid contact [14] Light and heat p>
5. Polymerization hazard[15] No polymerization
6. Decomposition products[16] sup> Hydrogen bromide
Storage method
Storage Precautions[17] Store in a cool, ventilated warehouse. Keep away from fire and heat sources. Keep container tightly sealed. They should be stored separately from oxidants, alkalis, and food chemicals, and avoid mixed storage. Use explosion-proof lighting and ventilation facilities. It is prohibited to use mechanical equipment and tools that are prone to sparks. The storage area should be equipped with emergency release equipment and suitable containment materials.
Synthesis method

Purpose
1. Used as solvent and in organic synthesis. [18]



 微信扫一扫打赏
微信扫一扫打赏
