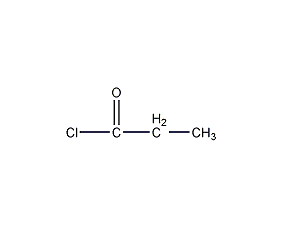
Structural formula
| Business number | 01P4 |
|---|---|
| Molecular formula | C3H5ClO |
| Molecular weight | 92 |
| label |
propionyl chloride, Pesticide intermediates; aliphatic carboxylic acids and their derivatives |
Numbering system
CAS number:79-03-8
MDL number:MFCD00000745
EINECS number:201-170-0
RTECS number:UG6657000
BRN number:385632
PubChem number:24898602
Physical property data
1. Properties: colorless liquid with pungent odor
2. Relative density (25℃, 4℃): 0.990670.9
3. Relative density (20℃, 4℃): 1.0646
4. Melting point (ºC): -94
5. Boiling point (ºC, normal pressure): 77~79
6. Refractive index at room temperature (n20): 1.4032
7. Refractive index: 1.4032
8. Flash point (ºC ): 12
9. Gas phase standard entropy (J·mol-1·K-1): 336.01
10 . Vapor phase standard hot melt (J·mol-1·K-1): 94.05
11. Vapor pressure (kPa, 25ºC): No Determined
12. Saturated vapor pressure (kPa, 60ºC): Uncertain
13. Heat of combustion (KJ/mol): Uncertain
14. Critical Temperature (ºC): Uncertain
15. Critical pressure (KPa): Uncertain
16. Log value of oil-water (octanol/water) partition coefficient: Uncertain
16. p>
17. Explosion upper limit (%, V/V): Uncertain
18. Explosion lower limit (%, V/V): Uncertain
19. Dissolution Properties: It is soluble in water and alcohol and decomposes quickly. It is soluble in benzene and ether.
Toxicological data
None yet
Ecological data
None yet
Molecular structure data
1. Molar refractive index: 20.82
2. Molar volume (cm3/mol): 86.5
3. Isotonic specific volume (90.2K ): 195.0
4. Surface tension (dyne/cm): 25.7
5. Polarizability (10-24cm3): 8.25
Compute chemical data
1. Hydrophobic parameter calculation reference value (XlogP): 1.2
2. Number of hydrogen bond donors: 0
3. Number of hydrogen bond acceptors: 1
4. Number of rotatable chemical bonds: 1
5. Topological molecular polar surface area (TPSA): 17.1
6. Number of heavy atoms: 5
7. Surface charge: 0
8. Complexity: 42.2
9. The number of isotope atoms: 0
10, The number of determined atomic stereocenters: 0
11, The number of uncertain atomic stereocenters: 0
12 , Determined number of stereocenters of chemical bonds: 0
13, Uncertain number of stereocenters of chemical bonds: 0
14, Number of covalent bond units: 1
Properties and stability
1. It is highly toxic and irritating, extremely irritating to the skin and mucous membranes, and even causes burns. It decomposes when exposed to water to produce hydrogen chloride. The production equipment should be sealed and the site should have good ventilation conditions. Operators must wear labor protection equipment.
2.This product is highly toxic and irritating. It can strongly irritate the skin and mucous membranes and even cause burns. It decomposes when exposed to water to produce hydrogen chloride. Production equipment should be sealed and the site should have good ventilation conditions. Operators must wear labor protection equipment.
Storage method
Packaging should be airtight, heat-proof and moisture-proof. Generally used as intermediate for self-use. Store and transport according to regulations on toxic chemicals. The packaging should be airtight, heat-proof and moisture-proof. Generally used as intermediate for self-use. Store and transport according to regulations on toxic chemicals.
Synthesis method
Propionyl chloride can be produced by reacting propionic acid with phosphorus trichloride, phosphorus pentachloride, thionyl chloride or phosgene.
1. Put propionic acid and phosphorus trichloride into the reaction pot, add several magnetic chips, and reflux at 50°C for 6 hours. Leave to stand for 1-2 hours, and then separate the lower layer of phosphorous acid to obtain propionyl chloride. Under the condition of propionic acid:PCl3 being 1.32:1, the yield is 95%. When the crude product needs to be purified, distillation can be used to collect the fraction at about 80°C to obtain the finished product. Raw material consumption quota: propionic acid (99.5%) 990kg/t, phosphorus trichloride (98%) 690kg/t.
![]()
2. Benzoyl chloride method It is obtained by the reaction of propionic acid and benzoyl chloride.
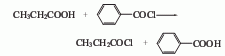
p>
Purpose
It is used as a propionylation reagent in organic synthesis and is an intermediate in the preparation of various propionic acid derivatives, such as phenylacetone, etc.; used in the production of the pesticide propanil; used in the pharmaceutical industry to produce the anti-epileptic drug diametal Because, cholerol, anti-adrenergic drug methoxyamine hydrochloride, etc.
Intermediates of the plant growth regulator prohexadione. In the pharmaceutical industry, it is used to produce anti-epileptic drug metoin, anti-adrenergic drug methoxyamine hydrochloride, and used as propionyl reagent in organic synthesis.



 微信扫一扫打赏
微信扫一扫打赏
